
Bioavailability
40% of drugs fail in preclinical trials due to bioavailability issues
As the average molecular weight of small molecules is increasing, there is a trend towards a higher share of low solubility molecules.
Improving bioavailability allows to reduce APIs dosage and minimize potential side effects.
Physical form (choice of the right salt, co-crystal, polymorph, amorphous) and morphology / size (particle engineering) need to be controlled in order to optimize solubility, dissolution rate, stability and powder flowability
Selection of functional excipients for galenic formulations will allow to further improve solubility and permeability.
Early evaluation of potential bioavailability issues
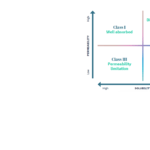
Solubility and permeability evaluation according to the Developability Classification System (DCS) for an early detection of potential bioavailability issues for oral drug candidates and solutions to be implemented.
- 40% to 60% of approved APIs are poorly soluble (classes II and IV)
- Most NCEs are in class II
Solutions to improve bioavailability
Screening and optimization of crystal structure activities
- Improve physico-chemical properties
- Polymorph screen (metastable form, hydrates and solvates)
- Salt formation (counter ion)
- Co-crystal formation (co-formers)
- Predictive tool XTAlpi (AI)
Particle size reduction
- Increase surface area
- Improve dissolution kinetics
Formulation
- Increase solubility and stability
- Enable controlled release
- Pre formulation (physico-chemical characterization)
- Excipients: catalogue and custom polymers
- Amorphous solid dispersion: amorphization techniques such as spray drying or hot melt extrusion

SEQENS bioavailability services to support each stage of drug development
1/ Early Development (Preclinical, Phase I and IIa)
- Selection of the most appropriate physical form
- Polymorph screening: mandatory for IND application
- Salt / co-crystals screening: improve physical properties in order to increase chance of success at preclinical phase
- Amorphous solid dispersion : improve solubility and stability
- API Characterization
- Crystallization Process development
- Particle size and solid dispersion engineering
Generation of Intellectual Property : New polymorphs or co-crystals allow to generate new patents and extend IP protection for the generics phase
2/ Late Development (Phase IIb, III)
- Comprehensive screening for an IP perspective
- In-depth API Characterisation
- Process optimisation
Why Choose Seqens ?
- Fully integrated (from preclinical to commercial supply) avoiding the risk of discontinuity in drug development
- Comprehensive integrated and standalone solid state offering
- Scientific and regulatory consulting expertise
- Agile service delivery model
Global R&D presence (US, EU, INDIA)