
Safety, Quality and Sustainable Innovation
Safety processes
The control of technological risk is our top priority. We are therefore committed to controlling the safety of our processes. This commitment is clearly defined in the Group’s EHS policy.
This commitment is central as the chemical and pharmaceutical industry must follow and comply with numerous regulations, in particular those on the prevention of major accidents and environmental protection.
- Seveso Directives in Europe;
- Industrial Emissions Directive (IED) in Europe;
- ATEX Directive in Europe;
- OSHA PSM (Process Safety Management) regulations in the United States;
- PSM regulations in China (China National Administration of Occupational Safety).
In order to guarantee a high level of safety on our sites, to reduce risks to the lowest possible level, and to comply with the various regulations, SEQENS deploys an EHS management system at its sites that integrates risk-based process safety.
The Group has deployed a mechanical integrity program which aims, among other things, to ensure that safety-critical components are identified and included in a robust inspection, testing and preventive maintenance plan.
- Achieve an annual process safety program completion rate in excess of 90%;
- Achieve an annual completion rate for the process risk analysis program(PHA) of over 90%;
- Continuously improve its mechanical integrity program to prevent loss of containment and the release of hazardous substances.
- 110% completion of precess hazard analysis program (PHA) by 2023
- The 90% target is met and exceeded, with 56 process risk analyses completed by 2023.
- The Billingham and Middlesbrough sites have further strengthened their safety practices, including pipeline integrity checks before each transfer of hazardous products.
- The Aramon site has improved the safety of its hydrogenation operations by installing a pneumatic powder loader under nitrogen.
- The Saint-Fons site has modified its vibrating screens to prevent any risk of dust explosion.
- The Neot Hovav site has installed a new fire protection system (sprinkler) in the workshop, as well as new safety devices at the anhydrous hydrochloric acid decanting station.
- The Roussillon site (Novapex company) has replaced its propane tank with a fireproof one.
- The Wuxi site has replaced its fire detection system.
Quality
Product quality and the conformity of associated activities are essential to SEQENS. In its Quality Policy, SEQENS is committed to ensuring that products are always delivered safely and reliably to customers, in compliance with applicable regulations and international standards.
This is especially true for the pharmaceutical products supplied by SEQENS. There can be no compromise on the quality of the active ingredients and excipients used in medicines.
- Manage activities through a comprehensive quality management system that complies with international standards, data integrity requirements, applicable regulations and standards (GMP17 , GDP18 , GxP19 , ISO);
- Remain constantly attentive to regulatory changes and customer expectations to guarantee customer satisfaction and develop new partnerships;
- Manage relations with our stakeholders and suppliers effectively and sustainably;
- Carry out our operations guaranteeing the quality and safe use of products to serve patients and consumers;
- Carry out operations guaranteeing the quality and safe use of products in order to serve patients and consumers;
- Continuously improve the efficiency and reliability of all our activities, in particular through digitalization;
- Develop staff skills, competencies, behaviors and effectiveness through individualized support and training, and encourage empowerment at work.
SEQENS’ objective is to ensure its customers 100% success in regulatory inspections and certification audits to ensure business continuity and to be recognized as a major and serious player in the field
In addition, all of the Group’s teams are doing everything possible to ensure customer satisfaction. SEQENS has set itself the objective of reducing the number of justified complaints by 20% between 2021 and 2025
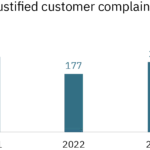
- Progress 2021-2023 : -+ 11%
- Target 2021-2025 : -20%
- In February 2023, the Roussillon site (Novapex) obtained its first Excipact certification. The site thus demonstrated that isopropanol was manufactured according to an adapted standard for excipients. This was the first example of its kind at this site.
- The Wuxi site passed its 1st GMP part 1 inspection in November 2023. This certification authorizes the sale of medicated intermediates in Europe.
- The Ecully site passed its 1st inspection on good distribution practices (GDP) to confirm the compliance of the distribution system for active ingredients manufactured at Novacyl sites in France, China and Thailand.
- The Villeneuve-la-Garenne site has deployed a Laboratory Information Management System (LIMS). This software package digitizes the main processes of the quality control laboratory. It manages, for example, field data entry and sample traceability, users, instruments, stocks and supplies, and product and equipment tracking. This system meets the quality and traceability requirements of customers and authorities.
Sustainable Innovation
Regulatory, safety and environmental issues require the development of efficient, reliable and sustainable production processes, from the design phase to the industrialization phase.
The Group relies on green chemistry to design and develop chemical products and processes that have minimal impact on the environment and human health, while being cost-effective.
Green chemistry focuses on innovative actions to prevent and reduce waste, develop the circular economy, optimize energy and raw material consumption with an emphasis on catalysis and enzymatic chemistry, take into account the demand for sustainable resources and replace hazardous substances (CMR).
SEQENS’ priorities for improving the sustainability of its products are :
- Renewable raw materials: introducing solutions based on plant-based raw materials into product manufacturing processes.
- Circular economy: internal reuse of used raw materials (co-products, by-products, waste) as inputs in the same or another production process.
- Green technologies: use a range of complementary in-house R&D solutions to minimize the environmental impact of products and processes, including biotechnology.
- Sustainable solutions: design and develop innovative solutions tailored to our customers’ key challenges, and support them in their quest for sustainable performance.
SEQENS has set itself the goal of evaluating 100% of its new proprietary products according to CSR criteria by 2025
The methodology for calculating the percentage of new proprietary products assessed according to CSR criteria was defined in 2023. The first CSR assessments will take place in 2024.
Propofol is a fast-acting intravenous anesthetic used for the induction and maintenance of general anesthesia and for sedation. The shortage of propofol during the Covid 19 crisis prompted a project to relocate the manufacture of this product to France. SEQENS has developed a manufacturing process for Propofol, which is interesting from a CSR point of view, since it uses flow chemistry and generates very little waste (vs. recent scientific publications). A patent application was filed in December 2021 and published in June 2023 to protect this new process. The site produced a demonstration batch of 4kg of propofol in 2023. A project to implement an industrial production unit is currently under study.