
A biocatalytic approach to improve the synthesis of Danoprevir
Focusing on biocatalysis for asymmetric synthesis and precious metal
catalysts replacement
The covid-19 crisis triggered an unprecedented mobilization of pharmaceutical research. Mid-April, 440 clinical trials were being conducted to identify a treatment, a large part of which being devoted to the search for antivirals.
To date, there is no universal vaccine capable of stimulating our immune system against coronaviruses. Synthetic chemical antivirals are the only weapon available for emergency use. If it is crucial to rapidly identify in clinical trials an antiviral effective on the Covid-19 target, it is equally important that the pharmaceutical production chain be organized in parallel to supply it. Reducing the development time of its synthesis route, as well as reducing its production cost, are major health challenges to respond to the emergency and ensure a wide distribution of the drug in this period of crisis.
An increasingly popular strategy for accelerating development is to reposition already commercially available compounds that are already approved on another therapeutic indications. Of the hundred or so small synthetic molecules currently being studied as antiviral treatments, approximately 10% are candidates for repositioning.
Danoprevir, a repositioned antiviral for Covid-19 treatment and a complex synthesis
Danoprevir is one such molecule. Discovered by Array BioPharma (now Pfizer) and first licensed to Roche for development and commercialization. Ascletis got a license for development and production in China where it is marketed since 2018 under the name Ganovo®. Danoprevir is a protease inhibitor used for the treatment of hepatitis C virus (HCV). The Ninth Hospital in Nanchang is currently conducting a clinical trial on a combination of danoprevir and ritonavir, to evaluate its potential for repositioning on COVID-19. The modified-peptidic type macrocyclic structure of Danoprevir is built according to a 20-steps convergent synthesis. The 3 main disconnections are made around 2 peptide bonds at the building block 1 and a ring-closure metathesis between building blocks 2 and 3.
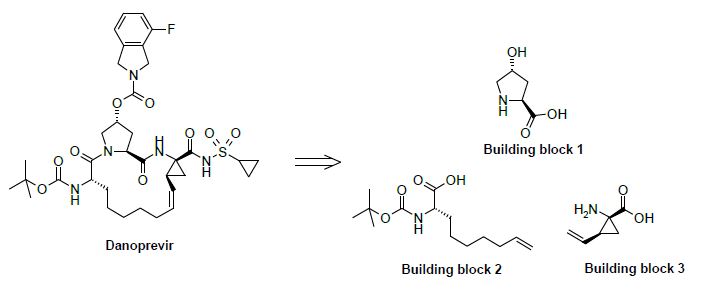
Building block 1: Asymetric synthesis using a Proline Hydroxylase
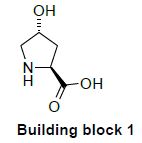
The hydroxy pyrrolidine analog is prepared by the direct hydroxylation of L-proline at position 4. Such a reaction at an inactivated position was made possible thanks to a L-proline-4-hydroxylase. This specific enzyme allows for an hydroxylation reaction with the unprotected proline, in mild conditions and with a perfect selectivity compared to what could be generally expected from a standard classical chemical synthesis.
This route clearly demonstrates the benefits of biocatalysis for the expedient synthesis of small complex chiral molecules, compared to a standard chemical route.
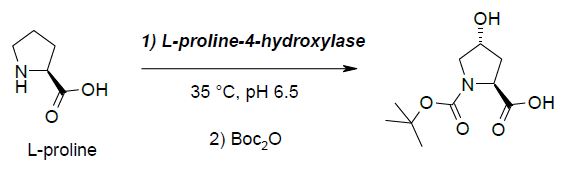
Building block 2: potential substituton of a chiral rhodium catalyst by an ene-reductase
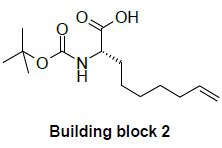
The unnatural α-amino acid derivative possessing a terminal alkene, is involved in a peptide bond with the hydroxy proline derivative and is one of the precursor for the ring closure metathesis. A straightforward synthesis of this compound relies on the Rhodiumcatalyzed asymmetric hydrogenation of the corresponding dehydroamino acid. Despite a perfect enantio- and chemo-selectivity for the conjugated alkene, the use of this transition metal catalyst suffers from a tedious preparation of the chiral diphosphine ligand and from the actual price of Rhodium, which has a commercial price that has increased by a factor 8 to 13 over the last few years.
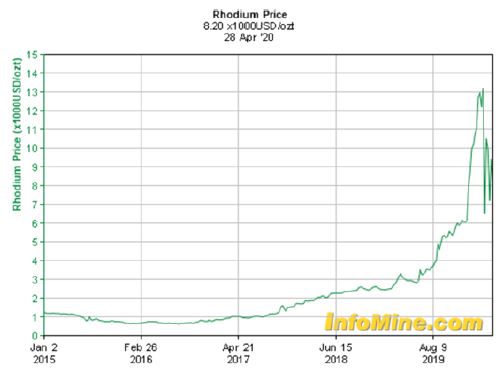
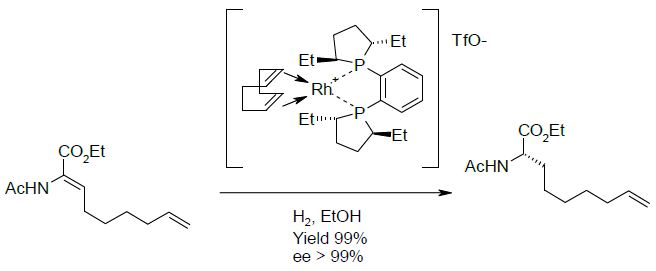
An elegant and cheaper alternative to this transition metal catalyzed hydrogenation would be the reduction of this enoate by an ene reductase (ERED).

This NAD(P)-dependent family of enzymes can reduce selectively substituted alkenes conjugated to an electron withdrawing group meaning that chemo-selectivity between the 2 alkenes could be achieved. Generally, the ERED-catalyzed reactions proceed with high enantioselectivity in mild conditions. An efficient system for the regeneration of the NAD(P)H cofactor could be easily implemented to afford a greener process. Furthermore, this biocatalytic reduction surrogate should allow for the reduced cost of the transformation.
First, enantio-selectivity relies on the enzyme and not on a chiral ligand requiring a multi-step asymmetric synthesis. Secondly, the reaction does not need any costly transition metal that has to be purged from the final product in order to avoid metal impurities and finally reach target specifications.
This family of enzymes (ene-reductases) is now part of Seqenzym®, enzyme kits proposed by “Protéus by Seqens” for fast assessment of enzymes activity on a target reaction.
The other precious transition metals (Palladium, Ruthenium) exhibited a similar sharp price increase during the last few years. Seqens is currently working for several of its customer projects on synthesis pathways for the replacement of precious metal catalysts by biocatalytic steps.
Biocatalysis is a key tool for more efficient and affordable drug synthesis
Protéus by Seqens applies a robust methodology for biocatalytic process development. First, we screen candidate enzymes from off-the-shelf enzymes, including said Seqenzym® kits, and a collection of exclusive sequences from a diverse strains library.
Once the best candidate enzyme has been identified, the two main development steps are the optimization of the biocatalytic process and the efficient production of the enzyme. Using Design of Experiments (D.O.E.) approach in both cases helps to reduce the number of tests and accelerate the development while studying and setting critical parameters.
Then, the biocatalytic process can be transferred to the Seqens CDMO manufacturing sites for the API synthesis, starting from grams to multi-tons.
When enzyme performances still need to be improved, Protéus by Seqens also proposes proprietary directed evolution strategies to enhance enzymes key features like activity or selectivity for instance.
The example of Danoprevir highlights the benefits of integrating biocatalytic steps within API synthesis. The synthesis of building block 1 demonstrates the interest of an enzymatic route for a chiral synthesis while the synthesis of building block 2 is an example of precious metal catalyst replacement by an enzyme.
With the world waiting for a treatment in order to efficiently tackle the Covid-19 pandemic, as soon as a drug will be identified, the next immediate challenge will be to quickly scale-up its production in order to make it available and affordable for a large number of patients.
An efficient synthesis will be required, if possible without the need for rare resources such as precious metals catalysts. Being dependent on precious and rare metals for such an essential need as Covid-19 drug synthesis is a risk, as it could exacerbate already existing supply tensions and become a limiting factor for widespread treatment usage. Biocatalysis is a differentiating asset and an alternative to address this challenge.
Seqens teams reviewed more than a hundred small molecules, and are ready to handle their development and industrialization, according to their stage of development. This race against time for the identification and development of a molecule against COVID19 is essential while awaiting the development of a vaccine. Thanks to its positioning in the drug value chain, Seqens is fully mobilized to bring its expertise and support pharmaceutical companies and emerging companies in their search for a treatment and in the production of APIs and pharma intermediates.
Authors :

Project Manager,
Protéus by Seqens, Seqens

Business Development Manager,
R&D Services, Seqens
More information about Proteus: www.proteus.seqens.com